СИНТЕЗ НОВЫХ 5-МЕТИЛСУЛЬФАНИЛАЗОЛО[1,5-<i>a</i>]ПИРИМИДИН-7(4<i>H</i>)-ОНОВ И ИССЛЕДОВАНИЕ ИХ ХИМИЧЕСКИХ И ЦИТОТОКСИЧЕСКИХ СВОЙСТВ
Ключевые слова:
кислота Мёльдрума, гетероциклизация, противоопухолевая активность, электрофильное замещение, азоло[1,5-a]пиримидиныАннотация
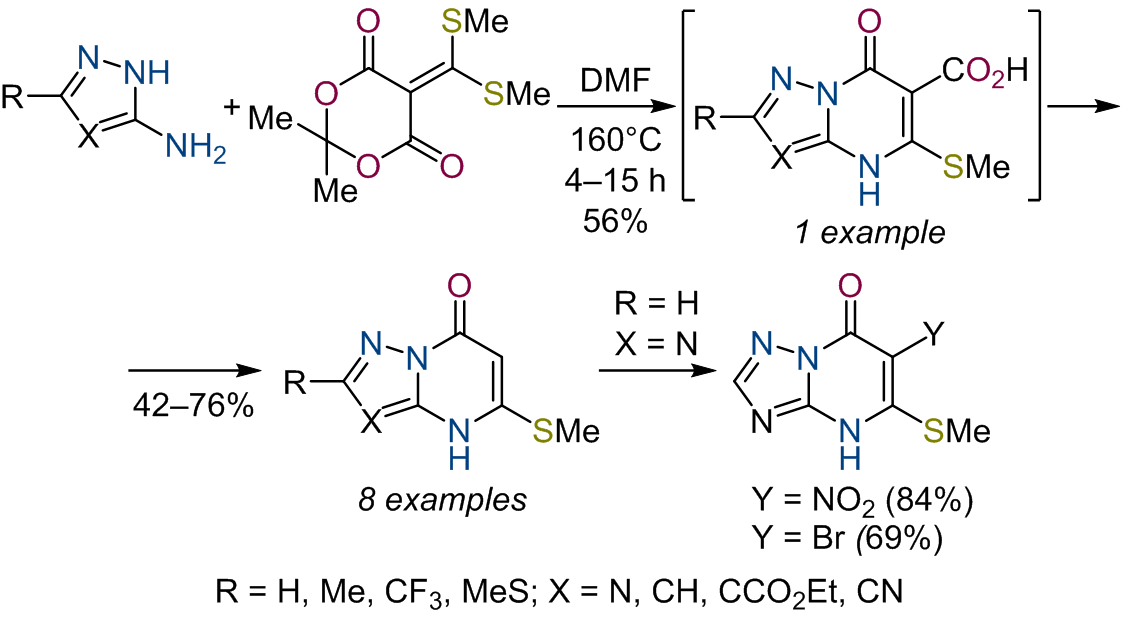
Разработан метод синтеза новых 5-метилсульфанилазоло[1,5-a]пиримидин-7(4H)-онов путем гетероциклизации 3-аминоазолов
и 5-[бис(метилсульфанил)метилиден]-2,2-диметил-1,3-диоксан-4,6-диона. При оптимизации процесса была выделена 5-метил-
сульфанил-7-оксо-4,7-дигидроазоло[1,5-a]пиримидин-6-карбоновая кислота, позволяющая сделать вывод о последовательности
превращениий реакции гетероциклизации. Исследована реакционная способность полученного ряда 5-метилсульфанилазоло-
[1,5-a]пиримидин-7(4H)-онов в классических реакциях электрофильного замещения. Проведена оценка цитотоксического
действия этих соединений в отношении опухолевых клеточных линий А549, HepG2 и RD и нормальных клеток HEK-293.